📋 Table of Contents
🔷 Introduction to Deterioration of Concrete
Deterioration of concrete refers to the progressive degradation of its physical, mechanical, or chemical properties over time, leading to reduction in strength, durability, aesthetics, and serviceability of the structure. Although concrete is perceived as a durable, maintenance-free material, it is susceptible to a wide range of deterioration mechanisms depending on its environment and quality of construction.
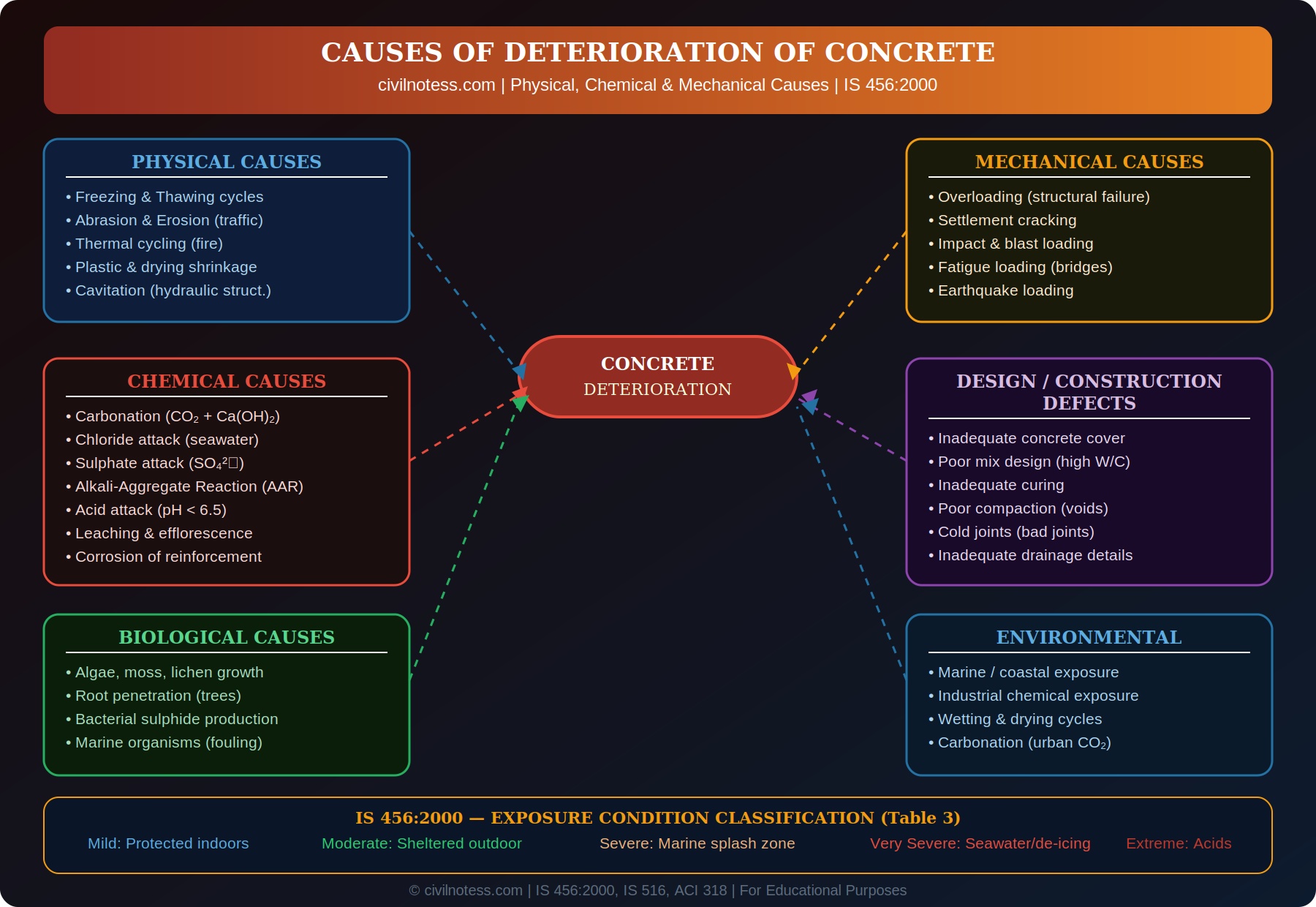
The causes of concrete deterioration can be broadly classified into:
- Physical causes — related to physical forces and environmental factors
- Chemical causes — involving chemical reactions with agents in the environment
- Mechanical causes — due to applied loads and structural forces
- Biological causes — caused by organisms
- Design and construction defects — poor quality of materials or workmanship
❄️ Physical Causes of Deterioration
1. Freezing and Thawing (Frost Action)
One of the most damaging physical mechanisms in cold climates. Water trapped in concrete pores expands by approximately 9% when it freezes. Repeated freeze-thaw cycles cause progressive internal cracking, surface scaling, and spalling of concrete. Particularly damaging to non-air-entrained concrete.
- Prevention: Air-entrainment (3–6% entrained air), low W/C ratio, adequate strength
- IS 456 requirement: Use of air-entraining agents recommended in frost-prone areas
2. Abrasion and Erosion
Mechanical wearing of the concrete surface due to rubbing or grinding:
- Abrasion: Wearing by rubbing contact — floors under heavy traffic (factories, car parks, warehouses)
- Erosion: Wearing by flowing water carrying suspended particles (hydraulic structures — spillways, channels, tunnels)
- Prevention: High-strength concrete (≥ M30), hard aggregate (granite), surface hardeners, steel fibre reinforced concrete
3. Thermal Changes and Fire
- Thermal cycling: Alternate heating and cooling causes differential expansion/contraction → fatigue cracking
- Fire damage: At 300°C, C-S-H begins to decompose; at 600°C, concrete loses ~50% strength; at 800°C+ concrete is essentially destroyed. Spalling (explosive) can occur at rapid heating rates due to steam pressure.
- Mass concrete thermal cracking: Core-to-surface temperature difference > 20°C causes thermal gradient cracks in large pours (dams, raft foundations)
4. Shrinkage Cracking
- Plastic shrinkage: Occurs within hours of placing when evaporation rate exceeds bleeding rate. Wide, shallow cracks on surface.
- Drying shrinkage: Long-term volume reduction as concrete dries. Causes flexural/tension cracking in restrained elements.
- Autogenous shrinkage: In low W/C, high-strength concretes — occurs due to self-desiccation even without moisture loss.
5. Cavitation
Occurs in hydraulic structures (gates, tunnel linings, spillways) where high-velocity water flow creates and collapses vapour bubbles. The collapse produces intense local pressures (up to 100 MPa momentarily) that pit and damage the concrete surface progressively. Very difficult to repair — prevention by hydraulic design (avoid flow separation) is key.
⚗️ Chemical Causes of Deterioration
1. Carbonation
Carbon dioxide (CO₂) from the atmosphere penetrates into concrete and reacts with calcium hydroxide:
Carbonation depth increases with time: x = k√t (x in mm, t in years, k = carbonation coefficient dependent on W/C and cover quality). Once carbonation reaches reinforcement level, rebar corrosion initiates even in the absence of chlorides.
2. Chloride Attack
Chloride ions (Cl⁻) penetrate concrete from seawater, de-icing salts, or admixtures and initiate localised (pitting) corrosion of reinforcement when the chloride threshold is exceeded:
- IS 456 threshold: 0.4% by weight of cement total chloride (for RCC)
- Chloride can be present from mixing water, aggregates, or external sources
- IS 456 limits: Max 0.4% Cl⁻ for reinforced concrete, 0.1% for prestressed concrete
3. Sulphate Attack
Sulphate ions (SO₄²⁻) in soil, groundwater, or industrial effluents react with cement hydration products:
- Ettringite formation: SO₄²⁻ + C₃A → ettringite (expansive) → cracking and disintegration
- Gypsum formation: SO₄²⁻ + Ca(OH)₂ → gypsum → softening of concrete paste
- IS 456 Table 4 classifies sulphate concentration and specifies cement type and W/C for each class
- Prevention: Use sulphate-resistant cement (SRC / IS 6909), low W/C, adequate cover
4. Alkali-Aggregate Reaction (AAR)
Alkalis in cement pore solution react with certain reactive aggregates (see dedicated post). Results in internal expansion, map cracking, and gel exudation. Prevention: limit alkali to < 3.0 kg/m³ Na₂O equivalent (IS 456 Cl. 5.4.1).
5. Acid Attack
Concrete has very poor resistance to acids (pH < 6.5). Acids dissolve Ca(OH)₂ and C-S-H gel, progressively disintegrating the concrete matrix:
- Industrial sewers (H₂SO₄ from bacterial oxidation of H₂S)
- Dairy/food industries (lactic acid, carbonic acid)
- Prevention: Acid-resistant coatings (epoxy, polyurethane), use of acid-resistant aggregates
6. Leaching and Efflorescence
Soft or aggressive water dissolves Ca(OH)₂ from concrete and carries it out of the matrix (leaching). The white deposits of CaCO₃ on concrete surfaces are called efflorescence. Long-term leaching creates porosity and weakens the matrix.
🔨 Mechanical Causes of Deterioration
- Overloading: Stresses exceeding design capacity → shear or bending cracks → progressive structural failure
- Settlement cracking: Differential settlement of foundations causes diagonal tension cracks in walls and beams
- Impact loading: Sudden high loads (blast, falling objects) cause localised spalling and fracture
- Fatigue: Repeated cyclic loading (bridges, industrial floors, offshore platforms) causes progressive crack growth even at stresses well below static failure load. Concrete fatigue limit ≈ 50–55% of static strength at 10⁷ cycles.
- Earthquake/seismic loading: Inadequate ductility detailing leads to brittle failure at joints
🌿 Biological Causes of Deterioration
- Algae and moss: Growth on moist surfaces. Hold moisture → promote carbonation and frost action. Roots penetrate cracks.
- Tree roots: Mechanical disruption of foundations and underground structures
- Bacteria: Sulphate-reducing bacteria (SRB) in sewers produce H₂S → oxidised to H₂SO₄ → severe acid attack on crown of sewer pipes. Biogenic sulphide corrosion (BSC).
- Marine organisms: Barnacles, mussels on marine concrete — hold moisture, accelerate carbonation and freeze-thaw damage
⚠️ Design and Construction Defects
| Defect | Consequence |
|---|---|
| Inadequate concrete cover | Early carbonation/chloride-induced corrosion of rebar |
| High W/C ratio | High porosity → easier penetration of aggressive agents |
| Inadequate curing | Incomplete hydration → low strength and high permeability |
| Poor compaction (honeycombing) | Voids create direct paths for aggressive agents to rebar |
| Cold joints (construction joints) | Planes of weakness and permeability — preferential attack paths |
| Inadequate drainage details | Water ponds on surfaces → leaching, freeze-thaw, carbonation |
| Wrong aggregate selection | AAR, excessive shrinkage, freeze-thaw vulnerability |
| Chloride-based admixtures | Direct initiation of rebar corrosion (CaCl₂ accelerators) |
🏭 IS 456 Exposure Classification (Table 3)
| Exposure Class | Environment | Min. fck (MPa) | Max W/C | Min. Cover |
|---|---|---|---|---|
| Mild | Protected indoors, no condensation | 20 | 0.55 | 20 mm |
| Moderate | Sheltered outdoor, high humidity | 25 | 0.50 | 30 mm |
| Severe | Exposed to sea water, alternate wetting/drying | 30 | 0.45 | 45 mm |
| Very Severe | Seawater, de-icing salts, aggressive ground | 35 | 0.45 | 50 mm |
| Extreme | Acid tanks, aggressive chemicals | 40 | 0.40 | 75 mm |
❓ Exam FAQs — Causes of Deterioration
Q1. What are the three essential conditions for chemical deterioration of concrete?
Most chemical deterioration requires: (1) A reactive agent (chloride, sulphate, CO₂, acid), (2) A transport pathway (permeable concrete, cracks, inadequate cover), and (3) Sufficient moisture (water acts as the transport medium for all ions). Remove any one of these three → deterioration stops or greatly slows.
Q2. Why is low W/C ratio the single most important factor in concrete durability?
Lowering the W/C ratio reduces capillary porosity (by filling more voids with hydration products), dramatically reducing permeability. Since most deterioration mechanisms require ions to transport through the concrete pore network (chlorides, sulphates, CO₂ dissolved in moisture), reducing permeability is the most effective single durability measure.
Q3. What is the IS 456 exposure classification for concrete in coastal areas?
Concrete in coastal areas (within 1 km of coast) subjected to sea spray is classified as Severe exposure. Concrete submerged in tidal/splash zones is Very Severe. These require min. fck = 30–35 MPa, max W/C = 0.45, min. cover = 45–50mm as per IS 456:2000 Table 3 and Table 16.
Q4. What is the carbonation depth formula and what factors affect carbonation rate?
Carbonation depth: x = k√t where x = depth (mm), t = time (years), k = carbonation coefficient. Factors increasing carbonation rate: high W/C ratio (high porosity), low cement content, inadequate curing, dry indoor environments (moderate RH 50–70% fastest), absence of silica fume/GGBS. Carbonation stops if concrete is continuously wet or continuously dry.
📝 Quick Summary — Causes of Deterioration
- Physical: Freeze-thaw | Abrasion | Thermal/fire | Shrinkage | Cavitation
- Chemical: Carbonation (CO₂) | Chloride | Sulphate | AAR | Acid | Leaching
- Mechanical: Overloading | Settlement | Impact | Fatigue | Earthquake
- Biological: Algae | Roots | SRB bacteria | Marine organisms
- Construction: Poor cover | High W/C | Bad compaction | No curing
- IS 456: 5 exposure classes (Mild → Extreme) → Table 3 & Table 16

