📋 Table of Contents
🔷 What is Alkali-Aggregate Reaction (AAR)?
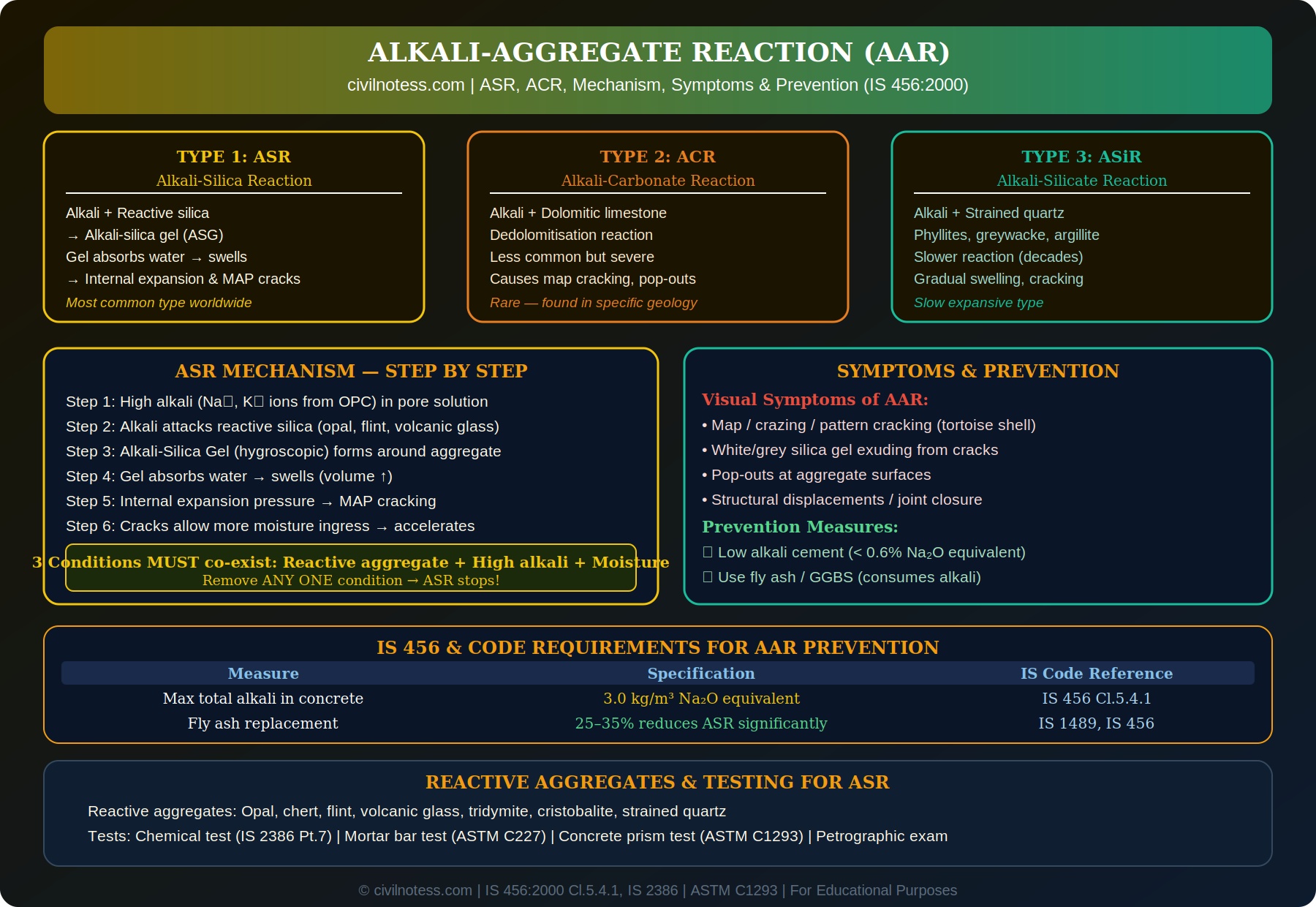
Alkali-Aggregate Reaction (AAR) is a deleterious chemical reaction that occurs within hardened concrete between the alkali hydroxides in the cement pore solution (derived from sodium and potassium oxides in cement — Na₂O and K₂O) and certain reactive minerals in aggregates. This reaction produces expansive products that cause internal pressure, cracking, and progressive structural deterioration.
AAR was first identified by Thomas Stanton in 1940 in California. It has since been discovered in concrete structures worldwide and is responsible for deterioration of many dams, bridges, and pavements globally.
🔬 Three Types of AAR
| Type | Full Name | Reactive Mineral | Frequency |
|---|---|---|---|
| ASR | Alkali-Silica Reaction | Opal, flint, chert, volcanic glass, amorphous silica | Most common globally |
| ACR | Alkali-Carbonate Reaction | Dolomitic limestone (clay-rich) | Rare — specific geology |
| ASiR | Alkali-Silicate Reaction | Strained quartz, phyllite, greywacke, argillite | Slow/gradual expansion |
1. Alkali-Silica Reaction (ASR)
The most common and well-studied form. Reactive amorphous or cryptocrystalline silica in aggregates reacts with alkali hydroxides in the pore solution:
The alkali-silica gel (ASG) formed is highly hygroscopic (water-attracting). It absorbs water from the concrete pore solution and swells. Since it is confined within hardened concrete, this swelling generates internal pressure that exceeds the tensile strength of concrete (~3 MPa), causing internal cracking radiating from the reactive aggregate particles.
2. Alkali-Carbonate Reaction (ACR)
Occurs with certain dolomitic limestones containing clay minerals:
Unlike ASR, ACR does not produce a gel. The expansion is caused by the swelling of clay minerals exposed by dedolomitisation. ACR is very aggressive and fast — no reliable preventive measures using SCMs (silica fume/fly ash) work for ACR. The only reliable prevention is to avoid reactive aggregates.
3. Alkali-Silicate Reaction (ASiR)
A slow-acting form involving strained or metamorphic siliceous materials (phyllites, greywacke, argillites). The reaction progresses over decades, causing gradual expansion and cracking. Often described as the “slow” or “late” ASR variant. Aggregates appear non-reactive in standard quick tests but show reactivity over long periods.
⚙️ ASR Mechanism — Step by Step
- Alkali source: Na₂O and K₂O in OPC dissolve in pore water → NaOH + KOH → high pH pore solution (12.5–13.5).
- Silica dissolution: OH⁻ ions from the high-pH solution attack and break the Si-O-Si bonds in reactive silica minerals at the aggregate surface.
- Gel formation: The dissolved silica combines with Na⁺/K⁺ and Ca²⁺ ions to form an amorphous alkali-silica-calcium gel (ASG) around and within the reactive aggregate.
- Water absorption: ASG is strongly hygroscopic — absorbs water from the pore solution. Swells significantly (osmotic pressure mechanism).
- Internal pressure: Swelling gel is confined by surrounding hardened cement paste and aggregate → builds up internal pressure (can exceed 10 MPa).
- Cracking: When internal pressure exceeds tensile strength of concrete → cracks initiate at aggregate-paste interface and propagate through cement paste.
- Positive feedback: Cracks allow ingress of more moisture → more gel swelling → more cracking → accelerating deterioration.
⚠️ Three Essential Conditions for AAR
🔑 The AAR Triangle — ALL THREE Must Be Present
1. Reactive aggregates — containing opal, amorphous silica, volcanic glass, dolomite, etc.
2. Sufficient alkalis — total alkali ≥ 3.0 kg/m³ Na₂O equivalent in the concrete
3. Moisture (water) — relative humidity ≥ 80% in the concrete pores
Remove ANY ONE of these three conditions → AAR stops completely.
This principle drives all AAR prevention strategies.
🪨 Reactive Aggregates for ASR
| Highly Reactive | Moderately Reactive | Potentially Reactive |
|---|---|---|
| Opal (amorphous silica) | Chert / Flint | Strained quartz |
| Natural volcanic glass (obsidian, rhyolite) | Quartzite (some) | Phyllite / Greywacke |
| Tridymite, Cristobalite | Sandstone (siliceous) | Argillite / Slate |
| Chalcedony | Granite (some) | Gneiss (some) |
Safe aggregates (non-reactive): Pure limestone, dolomite without clay, basalt, gabbro, quartzite (unstrained), river gravel (check petrography).
🔍 Symptoms / Indicators of AAR in Existing Structures
- Map cracking (crazing): Network of interconnected cracks in a polygonal tortoise-shell or map pattern on the concrete surface. Most characteristic symptom of ASR.
- White/grey silica gel exuding from cracks: Viscous gel deposits on crack surfaces and around aggregate particles. Gel may be white, colourless, or greyish.
- Closed/spalled expansion joints: ASR expansion pushes adjacent concrete elements together — joints designed for thermal movement close up and concrete crushes at joints.
- Structural displacements: Columns lean, beams deflect, dam faces bow outwards due to internal expansion.
- Pop-outs: Conical pieces of concrete pop out at the surface over reactive aggregate particles.
- Moisture-dependent cracking: Cracks are wider in wet weather (gel swells) and partially close in dry weather — distinctive pattern.
- Reduced mechanical properties: Compressive strength may remain near normal, but tensile, flexural, and shear strength are reduced significantly.
✅ Prevention of AAR (IS 456:2000)
Strategy 1: Limit Total Alkali Content
Use low-alkali cement (alkali content < 0.6% as Na₂O equivalent). The total alkali in concrete from all sources (cement + admixtures + water) must not exceed 3.0 kg/m³ Na₂O equivalent (IS 456 Clause 5.4.1):
Strategy 2: Use Supplementary Cementitious Materials (SCMs)
Replacing a portion of OPC with SCMs dramatically reduces ASR. SCMs work by:
- Fly ash (25–35% replacement): Consumes Ca(OH)₂ in pozzolanic reaction → reduces availability of alkali for ASR. Also reduces permeability.
- GGBS (40–50% replacement): Very effective — dilutes alkali in pore solution and produces a less permeable gel pore structure.
- Silica fume (10–15% replacement): Rapid pozzolanic reaction consumes Ca(OH)₂ → reduces pore solution pH and alkali → reduces driving force for ASR. Also extremely effective at reducing moisture ingress.
- Natural pozzolans: Similar mechanism to fly ash.
Strategy 3: Avoid Reactive Aggregates
Most reliable prevention — perform petrographic examination and chemical/mortar bar testing on aggregates before use. Avoid confirmed reactive aggregates. Where reactive aggregates cannot be avoided, use SCMs.
Strategy 4: Control Moisture
Keep concrete as dry as possible — waterproof membranes, sealers, and surface coatings. However, this is difficult to maintain long-term and should not be the primary prevention strategy.
🧪 Testing for AAR / Reactive Aggregates
| Test | Standard | Principle | Duration |
|---|---|---|---|
| Petrographic Examination | IS 2386 Pt.1 / ASTM C295 | Microscopic identification of reactive minerals | 1–2 weeks |
| Chemical Test | IS 2386 Pt.7 / ASTM C289 | Measures silica dissolved by NaOH | 24 hours |
| Mortar Bar Test | ASTM C227 | Mortar bars in humid cabinet — expansion measured | 3–6 months |
| Accelerated Mortar Bar Test | ASTM C1260 | Bars in NaOH solution at 80°C — rapid screening | 16 days |
| Concrete Prism Test | ASTM C1293 | Concrete prisms at 38°C, 100% RH — most reliable | 1–2 years |
| Uranyl fluorescence test | Research method | Identify ASR gel in thin sections under UV | Weeks |
ASTM C1260 (Accelerated Mortar Bar): Expansion < 0.10% at 16 days = non-reactive | 0.10–0.20% = uncertain | > 0.20% = potentially deleterious
ASTM C1293 (Concrete Prism): Expansion > 0.04% at 1 year = potentially deleterious
❓ Exam FAQs — Alkali-Aggregate Reaction
Q1. What are the three types of alkali-aggregate reaction?
1. Alkali-Silica Reaction (ASR) — most common, involves amorphous/cryptocrystalline silica minerals (opal, flint, volcanic glass). 2. Alkali-Carbonate Reaction (ACR) — involves dolomitic limestone, dedolomitisation mechanism, rare but aggressive. 3. Alkali-Silicate Reaction (ASiR) — involves strained quartz and metamorphic rocks, slow progressive expansion over decades.
Q2. What is the IS 456 limit for total alkali in concrete to prevent AAR?
As per IS 456:2000 Clause 5.4.1, the total alkali content from all sources shall not exceed 3.0 kg/m³ Na₂O equivalent, where Na₂O equivalent = %Na₂O + 0.658 × %K₂O. This is calculated based on the cement content and alkali content of the cement.
Q3. What are the three essential conditions for AAR to occur?
ALL THREE must be simultaneously present: (1) Reactive aggregates (opal, flint, volcanic glass, dolomite); (2) Sufficient alkalis (≥ 3.0 kg/m³ Na₂O equivalent); and (3) Moisture (RH ≥ 80% in concrete pores). Removing any one of these stops the reaction.
Q4. How does fly ash help prevent alkali-silica reaction?
Fly ash (25–35% replacement of OPC) prevents ASR through multiple mechanisms: (a) it dilutes the cement content, directly reducing total alkali per m³; (b) the pozzolanic reaction of fly ash with Ca(OH)₂ reduces the Ca(OH)₂ available to buffer pore solution alkalinity; (c) fly ash particles consume alkalis in pozzolanic reaction, reducing pore solution [OH⁻]; and (d) the denser gel pore structure reduces moisture ingress. Together, these remove or reduce all three essential conditions for ASR.
Q5. What is the characteristic visual symptom of ASR in concrete?
Map cracking (also called pattern cracking or crazing) — a network of interconnected cracks in a polygonal, tortoise-shell pattern on the concrete surface, often accompanied by white or grey silica gel exuding from the cracks. Pop-outs (conical spalls at aggregate locations) and joint closure (due to overall expansion) are also characteristic.
📝 Quick Summary — Alkali-Aggregate Reaction
- AAR = alkalis (Na⁺, K⁺) in cement + reactive minerals in aggregates + moisture
- Three types: ASR (most common) | ACR (dolomite — rare) | ASiR (slow)
- ASR reaction: SiO₂ + NaOH → Na₂SiO₃ (gel) → absorbs water → swells → cracks
- 3 essentials: Reactive agg. + High alkali (≥3 kg/m³) + Moisture (RH≥80%)
- Symptoms: Map cracking | Gel exudation | Pop-outs | Joint closure
- IS 456 Cl.5.4.1: Max 3.0 kg/m³ Na₂O equivalent
- Prevention: Low alkali cement | Fly ash/GGBS/SF | Petrographic testing | Sealers
